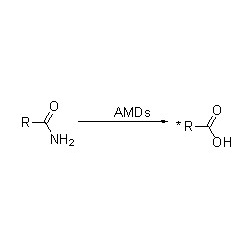
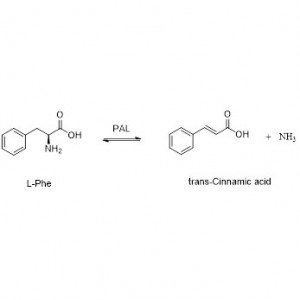
Amidase (AMD)
Ensaymyada:Waa kataliyayaal bayooloji oo macromolecular ah, inta badan enzymes-ku waa borotiinno.
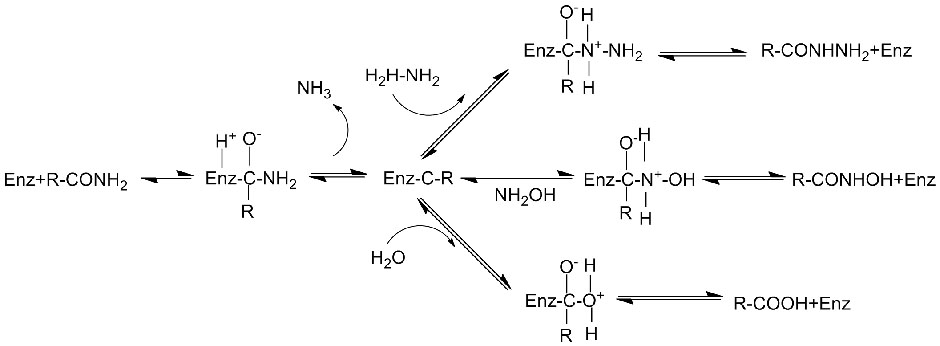
Amidase:Kordhi hydrolysis-ka noocyada kala duwan ee aliphatic iyo aromatic amides-ka ee gudaha iyo dibadda iyadoo loo wareejinayo koox acyl ah biyaha iyadoo la soo saarayo asiidhyo xor ah iyo ammonia. Asiidhyada Hydroxamic iyo asiidhyada kale ee dabiiciga ah ayaa si weyn loogu isticmaalaa daawooyin ahaan sababtoo ah waa qaybo ka mid ah arrimaha koritaanka, antibiyootikada iyo kuwa ka hortaga burooyinka. Amidases-ka waxaa loo qaybin karaa nooca R iyo nooca S acylases iyadoo loo eegayo stereoselectivity-ga kiciyaha.
Marka laga soo tago kicinta hydrolysis-ka amides, amidase waxay sidoo kale kicin kartaa falcelinta wareejinta acyl iyadoo ay joogaan substrates-ka isku dhafan sida hydroxylamine.
Amidase oo leh ilo kala duwan waxay leeyihiin gaar ahaansho substrate ah oo kala duwan, qaarkood waxay hydrolyze ku samayn karaan oo keliya amides-ka aromatic, qaarkoodna waxay hydrolyze ku samayn karaan aliphatic amides, qaarna hydrolyze α-ama ω-amino amides. Inta badan amines-ku waxay leeyihiin firfircooni catalytic ah oo wanaagsan oo keliya amides-ka acyclic ama aromatic fudud, laakiin aromatics-ka adag, amides-ka heterocyclic, gaar ahaan amides-ka leh substituents-ka ortho, guud ahaan firfircoonidoodu way yar tahay (insaymyo yar ayaa muujiya saameyn catalytic oo wanaagsan).
Habka kicinta:
| Ensaymyada | Koodhka Badeecada | Koodhka Badeecada |
| Budada Ensaymka | ES-AMD-101~ ES-AMD-119 | set ah 19 amidases, 50 mg midkiiba 19 shay * 50mg / shay, ama tiro kale |
| Qalabka Baaritaanka (SynKit) | ES-AMD-1900 | set ah 19 amidases, 1 mg midkiiba 19 shay * 1mg / shay |
★ Gaar ahaansho sare oo substrate ah.
★ Xulasho xooggan oo ku saabsan chiral.
★ Waxtarka sare ee beddelka.
★ Wax soo saar yar.
★ Xaalado falcelin fudud.
★ Deegaanka u wanaagsan.
➢ Baaritaanka ensaymka waa in loo sameeyaa substrate-yo gaar ah sababtoo ah gaar ahaanshaha substrate-ka, oo la helo ensaym kiciya substrate-ka bartilmaameedka leh saameynta katalytic-ga ugu fiican.
➢ Weligaa ha taaban xaalado daran sida: heerkul sare, pH sare/hoose iyo dareere dabiici ah oo leh xoog badan.
➢ Caadiyan, nidaamka falgalka waa inuu ku jiraa substrate, xalka kaydka (PH-ga falcelinta ugu fiican ee enzyme-ka). Substrates-ka sida hydroxylamine waa inay joogaan nidaamka falgalka wareejinta acyl.
➢ AMD waa in lagu daraa nidaamka falcelinta ugu dambeeya iyadoo la adeegsanayo pH-ga falcelinta ugu fiican iyo heerkulka.
➢ Dhammaan noocyada AMD waxay leeyihiin xaalado kala duwan oo falcelin ah oo ugu wanaagsan, sidaa darteed mid kasta oo ka mid ah waa in si gaar ah loo bartaa.
Tusaale 1aad(1):
Dhaqdhaqaaqa Hydrolysis-ka ee Substrates-ka Amid ee kala duwan
| Substrate | Hawl gaar ah μmols ugu yaraan-1mg-1 | Substrate | Hawl gaar ah μmols ugu yaraan-1mg-1 |
| Acetamid | 3.8 | ο-OH benzamide | 1.4 |
| Propionamide | 3.9 | p-OH benzamide | 1.2 |
| Lactamide | 12.8 | ο-NH2benzamide | 1.0 |
| Butyramide | 11.9 | p-NH2benzamide | 0.8 |
| Isobutyramide | 26.2 | ο-Tuluamide | 0.3 |
| Pentanamide | 22.0 | p-Tuluamide | 8.1 |
| Hexanamide | 6.4 | Nikotinamide | 1.7 |
| Cyclohexanamide | 19.5 | Isonicotinamide | 1.8 |
| Acrylamide | 10.2 | Picolinamide | 2.1 |
| Metacrylamide | 3.5 | 3-Phenylpropionamide | 7.6 |
| Prolinamide | 3.4 | Indol-3-acetamide | 1.9 |
| Benzamide | 6.8 |
Falcelinta waxaa lagu sameeyay xalka 50mM sodium phosphate buffer, pH 7.5, heerkul ah 70 ℃.
| Amides | Hydroxylamine | Haydraysiin |
| Acetamid | 8.4 | 1.4 |
| Propionamide | 18.4 | 3.0 |
| Isobutyramide | 25.0 | 22.7 |
| Benzamide | 9.2 | 6.1 |
Falcelinta waxaa lagu sameeyay xalka 50mM sodium phosphate buffer, pH 7.5, heerkul ah 70 ℃.
Fiirsashada walxaha la xiriira: amides, 100 mM(benzamide, 10 mM); hydroxylamine iyo hydrazine, 400 mM; ensaym 0.9 μM.
Tusaale 2(2):
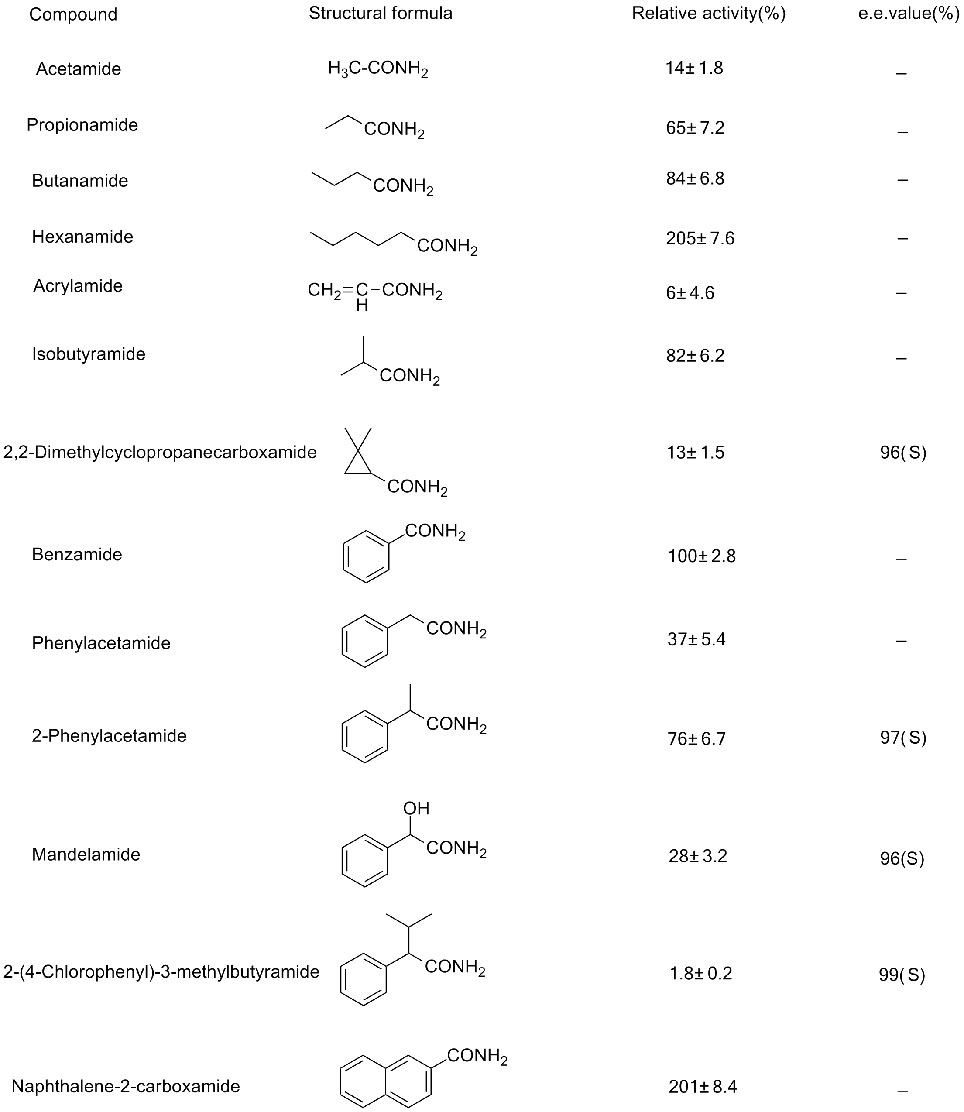
Tusaale 3(3):
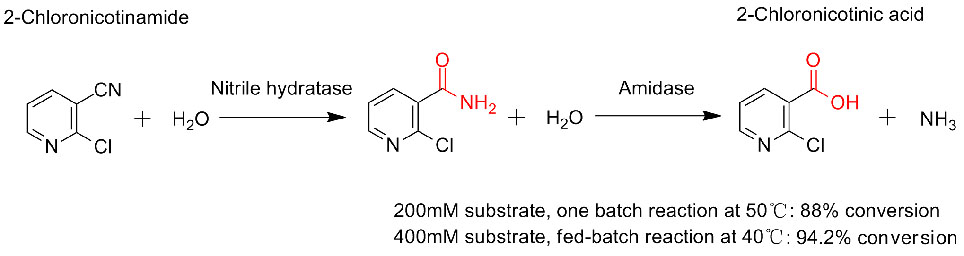
1. D'Abusco AS, Ammendola S., iyo al. Extremophiles, 2001, 5:183-192.
2. Guo FM, Wu JP, Yang LR, iwm. Process Biochemistry, 2015, 50(8): 1400-1404.
3. Zheng RC, Jin JQ, Wu ZM, iwm. Kimistariga Bayoorganik, 2017, Waxaa laga heli karaa khadka tooska ah 7.